While the earth remaineth,
seedtime and harvest,
and cold and heat,
and summer and winter,
and day and night shall not cease.
(Genesis 8:22)
One of the products of the continuing cycles of the seasons can be found on the bottoms of some lakes. Each spring, tiny plants bloom in Lake Suigetsu, a small body of water in Japan. When these one-cell algae die, they drift down, shrouding the lake floor with a thin, white layer. The rest of the year, dark clay sediments settle on the bottom. At the bottom of Lake Suigetsu, thin layers of microscopic algae have been piling up for many years. The alternating layers of dark and light count the years like tree rings. The sedimentation or annual varve thickness is relatively uniform, typically 1.2 mm per yr for present conditions in Lake Suigetsu which is located near the coast of the Sea of Japan. Recently scientists took a 75-m long continuous core from the center of the lake for close analysis including AMS 14C measurements on more than 250 terrestrial macrofossil samples of the annual laminated sediments.
----------
by H. Kitagawa, J. van der Plicht
"In order to build up a calendar time scale (i.e., varve chronology) for the Suigetsu (SG) core, a total of 85 subsamples were taken in a section of SG extending from 10.43 to 30.34 m below the top sediment, each ca. 25 cm in length, including a 1.5 cm overlap with neighbouring subsamples. To allow detailed observation of the sediments, the well-cleaned surfaces of sediments were scanned with a digital camera. By means of computer image analysis of digital pictures, we found that the much less distinct varves observed in some intervals during the deglaciation and Glacial could be determined only with a relatively large error. In order to reconstruct a more precise and longer varve chronology for the laminated sediments from Lake Suigetsu, we have reassessed the varve chronology in the whole section during the deglaciation as well as the Glacial up to a depth of 30.45 m.
The uncertainty in the varve chronology comes from two sources: core sampling and varve counting. Detailed comparison with short piston cores shows that the sampling does not cause significant loss of varves - typically 0-2 cm to a maximum of 3 cm, corresponding to ca. 20-30 yr in the Holocene and ca. 50 yr in the Glacial. Since the varve ages from below 18 m (corresponding to ca. 20,000 cal BP) were estimated by varve counting of a single core, the ages quoted should be considered as minimum ages, the error increasing with depth. Based on the results of some duplicated countings of selected subsamples and independent counting of different subsamples collected from the same horizon, we estimate that the counting error is less than 1.5%, corresponding to 150 yr for 10,000 varve years.
From the laminated sediments we selected terrestrial-origin macrofossils such as leaves, branches and insects for AMS 14C measurements. (see Appendix B) The samples are processed using a strong acid-alkali-acid (AAA) treatment for both samples and reference blank materials. The reference blank consists of more than 50 14C-free plant materials collected from the deep layer of the same core (corresponding to an age of ca. 90-100 ka). The Lake Suigetsu floating varve chronology consists of 29,100 varves. The sedimentation or annual varve thickness is relatively uniform (typically 1.2 mm yr-1 during the Holocene and 0.62 mm yr-1 during the Glacial). The age below 30.45 m depth is obtained by assuming a constant sedimentation in the Glacial (0.62 mm yr-1).
In order to reconstruct the calendar time scale, we compared the Lake Suigetsu chronology with calibration curves obtained from recently revised absolute German oak and the floating German pine calibration curves (2). Figure PE-4 shows the best match between the tree-ring and the Lake Suigetsu chronologies, estimated by minimizing the weighted sum of squared differences between the 14C ages of macrofossils and the tree-ring calibration curve. The features in our data overlapping the tree-ring calibration agree very well, even for "wiggles" in the 14C calibration curves. Using this match, we defined the absolute time scale for the Lake Suigetsu varves chronology. The 29,100 yr Lake Suigetsu chronology then covers the absolute age range from 8830 to 37,930 cal BP.
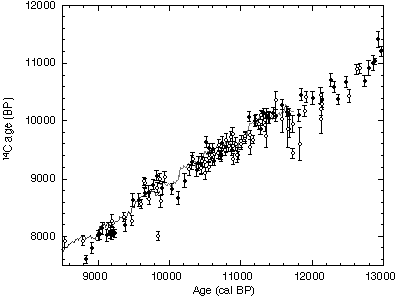
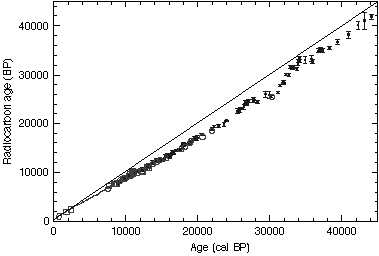
The combined 14C and varve chronologies from Lake Suigetsu are used to calibrate the 14C time scale beyond the range of the absolute tree-ring calibration. Figure 2 shows an atmospheric 14C calibration for the complete 14C dating range (less than 45 ka).The tree-ring calibration range, our calibration agrees well with the European sediments (3) and generally with marine calibrations obtained by combined U/Th and 14C dating of corals (4,5). ...
----------
The results from just one source could possibly be readily contested, but in this case the scientists have correlated the results from multiple sources including that of Lake Gosciaz (Poland), German oak and pine tree ring chronologies and also calibrations from coral data. Many in the scientific community are proposing the result of the above study as a "calibration" to radiometric C14 data, see Appendix A. Also the data seems to indicate no more that a 16.7 percent error due to deviation of C14 in the atmosphere for the past 40,000 years.

The above Table 2 from Palaeo 122(1996)p.114 summarizes published data concerning one proposed event, the termination of the Younger Dryas period, and illustrates the range of dating errors from various sources including tree rings, lake varves, coral dating and the Greenland ice cores. At the extremes there is a range from 10,447 to 11,700 year B.P.. The average being 11,074 yr B.P. plus or minus 627 years, a plus or minus 5.7 percent error range.
Conclusion: The apparent close correlation of the dating results from multiple
sources appears to be strong evidence for an earth much older than 10,000 years!
And
evidence that properly conducted C14 radiometric dating can approach reasonable
accuracy, possibly within better than 10 percent.
Also C14 dating affirms Scripture/Scripture affirms C14 dating!




Appendix A: The Radiocarbon age scale vs the 'real' (calibrated) years age scale.
from http://www.esd.ornl.gov/projects/qen/nerc14C.htmlMost information on the past 30,000 years or so is from sites or specimens that have been dated using radiocarbon (14C). However, the radiocarbon age scale that would be calculated from first principles (based on the decay rate of the 14C isotope, assuming that 14C was at the same level of abundance as it is at present) is not always reliable, because there have been fluctuations in the rate of production in 14C at the top of the atmosphere. The problems are particularly great at about 10,000 14C y.a., when a large influx of 14C-depleted carbon from the oceans, combined with a decrease in the rate of 14C production at the top of the atmosphere, gives an 'age plateau' such that the same 14C age covers a wide span of real time, about 1,000 years. Other dating methods (e.g. U/Th) can be used to attempt to check the 'true' age of specimens or sediment layers dated by 14C, although these all have substantial error margins of their own. The most convincing way to check the 14C age scale is through biological or sedimentological features which build up annual layers over long periods of time (e.g. tree rings, and annual layers of sediment building up on lake beds); counting back the annual layers will reveal the true number of years before the present, and comparing the 14C age of each tree ring or sediment layer will give an age scale for how 14C age can be converted into 'real' age. However, even this method is not completely reliable; 'false' double rings can sometimes appear, and occasionally a year may not appear in the record. Because of these problems, individual ring or layer-counting studies often suggest 'real' ages differing from one another by several percent, though they all suggest that the 'real' age is older than the 14C age before about 3,000 years ago. The most recent working consensus (adopted by papers in leading Quaternary journals e.g. Dahl & Nesje 1996) 14C-to-real age conversion scale is given below, but because it is possible that opinions on the appropriate age conversion will change as more data come in, the time slices of the maps are presently described according to a 14C age scale. The reader can use this preliminary age scale as a guide to the likely true age of each of the time slices and vegetation distributions given on this page and on the QEN Pages. Useful sources on the current understanding of the radiocarbon timescale include Kilian et al. (1995), Goslar et al. (1995) and Stuiver et al. (1993). For times before 12,000 14 y.a., the table is mainly based upon the multi-source curve published by Kitagawa & van der Plicht (1998).
It is necessary to bear in mind that quite apart from all the problems of calibration, a significant proportion of radiocarbon dates are not reliable for any purposes, because they have been contaminated with older or younger carbon that changes the apparent age of the sample. Many radiocarbon-dating specialists still refer to their field as 'more an art than as science'! Published radiocarbon dates from sites and layers of fossils and sediments are quite often rescinded, when the materials are found to have been naturally contaminated. Most often the contamination is from older (less 14C-rich) calcium carbonate, coal or charcoal washed in from other layers, making a sample or layer seem older than it actually is. Although radiocarbon dating is a very useful tool for the Quaternary palaeoecologist, it must always be interpreted with caution.
14C years ago=>Calibrated ('real') years ago
1,000 => 1,000
2,000 => 2,000
2,500 - 2,800 => 2,600
(sudden shift in atmospheric 14C content)
3,000 => 3,200
4,000 => 4,500
5,000 => 5,900
6,000 => 6,950
7,000 => 7,900
8,000 => 8,900
9,000 => 10,000
10,000 => 11,200 - 12,200
('radiocarbon plateau')
11,000 => 12,900
12,000 => 14,000
13,000 => 14,500
15,000 => 17,000
16,000 => 19,500
17,000 => 21,000
18,000 => 22,500
20,000 => 24,500
25,000 => 28,000
30,000 => 35,000
40,000 => 45,000
Appendix B: CHILL-10,000 sampling protocol for AMS radiocarbon dating
by
Hilary H. Birks & André F. Lotter
from http://www.cx.unibe.ch/sgi/Dating.htm
In contrast to conventional radiocarbon decay-counting, AMS radiocarbon dating requires sample sizes that are substantially smaller. Generally, a sample size of between 1-2 mg organic carbon is needed for AMS radiocarbon dating. This varies between laboratories, so it is wise to check with your laboratory before proceeding. Besides the advantage of allowing a better stratigraphic resolution (i.e. slices of 0.5-1 cm instead of e.g. bulk sediment samples using 5-10 cm of the core) we have also to take into consideration that when dealing with such small samples a small amount of contamination will lead to a substantial deviation of the 'true' radiocarbon age. Therefore, Schumacher's (1973) approach should not be followed here.
The standard procedure of sample preparation adopted by the different CHILL-10,000 projects should follow the protocol listed below:
Separation of terrestrial plant macrofossils from the sediment
1. Cut the wet core into suitable slices. Avoid sampling slumps or turbidites as they may include older, reworked material.
2. Do not store these samples more than a few weeks in the refrigerator (see Wohlfarth et al., 1998). If you cannot proceed immediately, it is best to deep-freeze the samples.
3. Sieve the sample through a 250 mm mesh. NB, if macrofossil analysis is to be done on the sample(s), sieve through a 125 mm mesh to retain small seeds etc.
4. If the sediment will not break down immediately, soak it under water in a clean beaker with a lid. If necessary, sediment breakdown can be assisted by adding a small teaspoon of tetra-sodium diphosphate - 10-hydrate crystals Na4P2O710H2O, a.k.a. sodium pyrophosphate, or Calgon water softener. Shake to dissolve the powder and stand overnight or as long as necessary in the refrigerator. If the sediment is calcareous, treatment with 10% HCl may be desirable. If it is humified peat, treatment with 10% KOH may be desirable.
5. Sieve the sample. It may be useful to use a selection of mesh sizes. If macrofossil material is sparse, save the sediments >125 mm in addition to the larger fractions.
6. Store the sieved material under water in clean beakers with lids, both clearly labelled, in the refrigerator. If you cannot proceed immediately, deep-freeze the samples.
Selection of material for dating
1. Disperse a little of the material in water in a clean shallow (petri) dish. Under a stereo-microscope, magnification ca. 12x, pick out the plant fragments with soft (entomological) forceps. It may be useful to separate the different types, e.g. mosses, leaves, seeds, etc. for easier identification, or selection of one sort of material. The material for dating should be identified.
2. Remove any adhering sediment or other material with a small brush or forceps.
3. Under the microscope, pick out the remains once more, carefully avoiding sediment and any dust, hairs etc. and place them in water in a new clean container. This is a washing process.
4. Pick out the remains again and place them in a dry, dust-free box or glass. Let the sample dry, covered, at room temperature or at ca. 50oC in an oven overnight.
5. Weigh the sample to ensure you have enough material for dating, as required by your dating laboratory. Although some laboratories can use as little as 2 mg dry material, to produce sufficient carbon it is usually best to submit at least 12 mg. For d13C measurements, 20 mg is required.
6. Control the dry sample once more for dust or clothing hairs. Wool hairs for example will make the sample 'younger'; synthetic oil-derived hairs will make the sample 'older'. With such small samples it is very important to do this.
7. Sterilize a glass vial at 600o C for 3 minutes. Place the clean sample in it. Label both the vial and the lid. Glass containers are preferable, as plastic suffers from static electricity which makes the fragments jump about.
8. The dry samples can be stored as necessary in a dark cold-room until being sent to the laboratory.
Steps 1-4 can be carried out in a laminated air flow chamber to reduce the chances of contamination by air-borne dust and clothing hairs. Important: As AMS samples are very small, already small amounts of fungal or bacterial biomass may easily contaminate the samples. Therefore, care should be taken to avoid this at all stages in the preparation; by proceeding rapidly, and by storing the samples in a dark refrigerator between stages. If delay is unavoidable, the material should be deep frozen. Samples should never be left in water at room temperature, as fungal growth can be extremely rapid. Samples that have been stored in this way, or in organic liquids (e.g. alcohol, glycerol etc.) should not be used for dating or d13C measurements. Working conditions should be as clean as possible, and care should be taken to remove all dust, hairs, etc from containers before use, and to control the samples for these contaminants as outlined above. With larger fragments, such as wood, do not handle them with your fingers.
Before sending off the samples for dating, note the following data in your lab journal:
- lake
- core ID, segment, date of coring
- core depth (relative and absolute) of sample
- date of macrofossil processing, chemical treatment
- what plant material is used for dating (e.g. 5 Abies needles, 20 Betula fruits, etc.)
- dry weight of submitted material
- expected age
When reporting the radiocarbon ages in publications, the following information should be given (see Stuiver & Polach, 1977):
- sample depth or code
- dating lab sample number
- material dated
- radiocarbon age ±1sd
- calibrated age (as cal years BP)
- d13C if measured
For calculating accumulation rates for biotic and abiotic variables within the framework of CHILL we suggests using a chronology that is based on a calendar age-scale. Calibration of radiocarbon ages can be done with various programs. The most commonly used is CALIB 3.0 (Stuiver & Reimer, 1993). This software can be down-loaded from the WWW (http://depts.washington.edu/qil/). Caution should, however, be used when calibrating late-glacial radiocarbon ages (i.e. >10,000 14C BP), because this software uses the coral-derived U/Th dates that as yet are sparse, and not verified yet by independent chronologies. The next version, CALIB 4.0, will be available at the end of 1998. It uses the revised Hohenheim dendrochronology, and for ages >10,000 BP, it incorporates more coral U/Th measurements and the laminated Cariaco Basin data back to ca. 14,500 14C BP (Hughen et al., 1998). In addition, a new calibration data set from Lake Suigetsu, Japan, is now available back to ca. 40,000 14C BP (Kitagawa & van der Plicht, 1998). This chronology is based on AMS dates of terrestrial plant material from annually laminated lake sediments (see Radiocarbon 40, 1998, and the calibration issue of Radiocarbon to be published in late 1998).
References
Hughen, K.A., Overpeck, J.T., Lehman, S.J., Kashgarian, M., Southon, J.R. & Peterson, L.C. 1998. A new 14C calibration data set for the last deglaciation based on marine varves. Radiocarbon 40: 483-494.
Kitagawa, H. & van der Plicht, J. 1998. A 40,000-year varve chronology from Lake Suigetsu, Japan: extension of the 14C calibration curve. Radiocarbon 40: 505-515.
Schumacher, E.F. 1973. Small is Beautiful: Economics as if people mattered. Blond and Briggs.
Stuiver, M. & Polach, H.A. 1977. Discussion. Reporting of 14C data. Radiocarbon 19: 355-363.
Stuiver, M. & Reimer, P.J 1993. Extended 14C data base and revised 14C calibration program. Radiocarbon 35: 215-230.
Wohlfarth, B., Skog, G., Possnert, G. & Holmquist, B. 1998. Pitfalls in the AMS radiocarbon-dating of terrestrial macrofossils. Journal of Quaternary Science 13: 137-145.
28.October 1998