Life from Dead Stuff?
"The uniform failure in literally thousands of experimental attempts to synthesize protein or DNA under even questionable prebiotic conditions is a monument to the difficulty in achieving a high degree of information content, or specified complexity from the undirected flow of energy through a system. We must not forget that the total work to create a living system goes far beyond the work to create DNA and protein...
"The probability that at ordinary temperatures a macroscopic number of molecules is assembled to give rise to the highly ordered structures and to the coordinated functions characterizing living organisms is vanishingly small. The idea of spontaneous genesis of life in its present form is therefore highly improbable, even on the scale of billions of years during which prebiotic evolution occurred."
"No matter how large the environment one considers, life cannot have a random beginning ... there are about two thousand enzymes, the chance of obtaining them all in a random trial is only one part in (10^20)^2000 = 10^40,000 , an outrageously small probability that could not be faced even if the whole universe consisted of organic soup."
Furthermore, no geological evidence indicates an organic soup, even a small organic pond, ever existed on this planet. It is becoming clear that however life began on earth, the usually conceived notion that life emerged from an oceanic soup of organic chemicals is a most implausible hypothesis. We may therefore with fairness call this scenario "the myth of the prebiotic soup.""
SOURCE: Thaxton, Charles B., Bradley, Walter L. & Olsen, Rodger L.
The Mystery of Life's Origin; Reassessing Current Theories
With the apparent collapse of the organic soup theories, hydrothermal vents
on the ocean floor began to play a important role in most origin-of-life explanations.
Penn State and SUNNY-Stony Brook researchers recently evaluated the likelihood of
ammonia formation (NH3) under primitive hydrothermal vent conditions.
Ammonia serves as a key starting material in the synthesis of amino acids and other
biologically important nitrogen-containing compounds.
"Reduced nitrogen compounds (e.g., NH3
(NH4+), HCN, and Urea) are far more reactive
reagents in prebiotic synthesis reactions than N2.
Hence, for prebiotic synthesis reactions, such as amino acid
formation, to occur in and around hydrothermal systems reduced
N-compounds need to be advected or formed in situ. Here we
have examined the in situ formation of reduced N-compounds.
Reaction path modeling indicates that ammonium is the stable
N-species below 350 °C in solutions buffered by any of the
common mineral buffers, except hematite-magnetite. None of the
conditions yield any other reduced N-compounds at
concentrations above 10-9 M. The few available gas
data on modern vent systems show, however, that N2
is far more abundant than NH4+. Possible sources
for N2 include unreacted N2 dissolved in
recharging seawater or mantle-derived N2. We
compared molar N:2/Ar ratios for vent solutions to those for
MORB (65) and air-saturated seawater (asw) (36). Furthermore,
we compared the concentration of dissolved N2 in
vents to that in asw. The data suggest that most N2
entrained in the recharging seawater does not react.
Therefore, to provide reduced N-compounds for prebiotic
synthesis in and around vent systems it is necessary to: 1)
advect the reduced compounds or 2) have a more reactive
mineral assemblage on the early earth."
This lack of adequate ammonia production under the conditions tested makes the origin of life at deep-sea hydrothermal vents unlikely. Also there are a number of other inhibiting factors including "new evidence indicates that transition metals and rare earth compounds in the early earth's oceans would have promoted the decomposition of what many scientists believe were key intermediate chemical compounds taking part in the most widely accepted evolutionary origin of life scenarios"

Reference: Martin Schoonen, "Reduced Nitrogen Compounds in Hydrothermal Vent Systems: A Critical Evaluation" SUNY-Stony Brook, Oral Presentation, 17 April 2002 Origin of Life/Prebiotic Chemistry Conf: Mitsuhiko Akaboshi et al., "Dephosphorylating Activity of Rare Earth Elements and Its Implication in the Chemical Evolution", 12th intl. conf. on the origin of life, San Diego, Ca.,1999
Well then, how about "panspermia", which says that life on Earth got its start in dormant life arriving from space. At the 32nd annual Lunar and Planetary Science Conference, Dr. H. Jay Melosh, from the University of Arizona, reported on a study that tested the probability of just one aspect of the meteor-transport hypothesis. The study reveals that Earth captures only about 3x10-16 interstellar meteor per year (one every three quadrillion years) and about 1x10-12 per year (one every trillion years) in a process by which an interstellar meteor is first captured by Jupiter and then eventually lands on earth. Translation: Odds are less than one in 200 that Earth would capture even one such meteor in its entire history. Then the odds grow even worse when one considers the narrow window of time in the life history of the Earth within which life was "seeded" on the Earth. The odds then grow to more than one in 5000, and that particular meteor has to have on board the very special "dormant seed" that has survived the deadly ubiquitous cosmic rays and is the right seed at the right time to begin primitive life on Earth. Undoubtedly many more studies will follow, but the outcome seems rather predictable. People who had expected panspermia to solve the problem of the rapid origin of life on Earth will have to look elsewhere!
(summary of "Intersteller Rocks Miss the mark" by Roger Wiems, FFF Quarter 2 -2001, issue 6, p 8)
Hot off the presses comes the latest theory concerning the origin of life!
"Life on Earth may have begun in rocks on the ocean floor. More than 4 billion years
ago, tiny cavities in minerals may have served as the first cells, two biologists
are proposing.
Iron sulphide honeycomb: a
fertile environment.
©
Canup/Asphaug
The key to the new theory is iron sulphide. Hot springs deposit a honeycomb of
this mineral on the ocean floor, with pockets a few hundredths of a millimetre
across. This would have been the ideal place for life to get going, say
William Martin, of Heinrich-Heine University in Dusseldorf, Germany, and
Michael Russell of the Scottish Universities Environmental Research Centre in Glasgow, UK.
"There are lots of theories [of the origin of life] but ours is the first
to start with the cell," says Martin. Most scientists assume that self-replicating
molecules or proteins came first.
Iron sulphide speeds up the reactions that join inorganic molecules together
into organic ones - some bacteria still use it. The hot water flowing out of
springs and into the honeycomb's holes is rich in the raw ingredients for
these reactions, such as ammonia and carbon monoxide. Bacteria can use such
compounds as fuel.
Confined to iron sulphide cells, simple compounds could become concentrated,
sparking the creation of complex molecules such as proteins and genetic
material.
|
Other scientists agree that iron sulphide honeycombs are fertile environments, but point to the gap between simple organic reactions and the chemistry of life. "The main problem is to explain how, where and why molecules such as enzymes emerge," explains biochemist Pier Luigi Luisi of the Federal Institute of Technology in Zurich, Switzerland."
( from http://www.nature.com/nsu/021202/021202-3.html "New theory for origin of life, Mineral cells might have incubated first living things." 4 Dec 2002 )The UNIQUENESS OF our LIFE SYSTEM:
AGE: Famed researcher Manfred Eigen headed a team that estimated
the age of the genetic code at 3.8 +- 0.6 billion years.(1)
The latest geochemical evidence places life's first
appearance on Earth at 3.86 billion years. In other words,
the origin of the genetic code coincides with the origin
of life.
If true, this rules out the origin of life having a
source other than on our Earth.
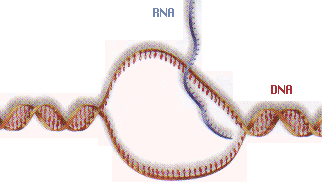
SURVIVAL: Even the simplest forms of life depend upon the
symbiotic relationship among three very complex sets
of molecules, DNA, RNA, and proteins. Some had theorized
that the first life was based upon a single set of
molecules termed "super RNA". Experiments with RNA indicated
that RNA molecules and even the nucleotides on which they
are built were chemically unstable under all possible
early earth environments.
The only place RNA molecules are safe from
chemical destruction is inside the membrane walls of the cell.(2)
RNA cannot survive without cells and cells cannot
survive and multiply without RNA. They could not have
originated independently, or one before the other.
If true, this rules out chance linkages of nucleotide sections
in either a prebiotic soup or at hydrothermal vents as the source of life.
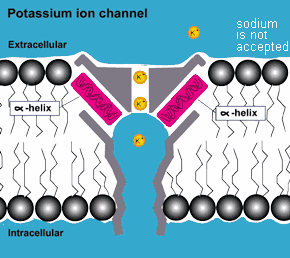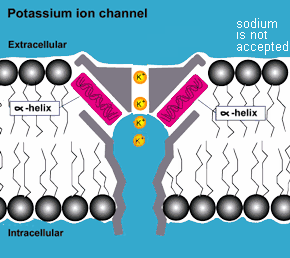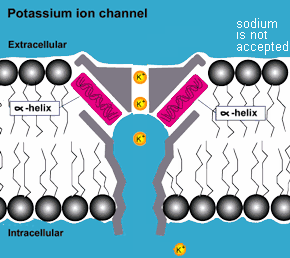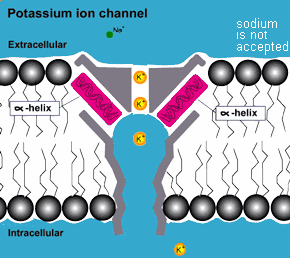
COMPLEXITY: In addition to the protective environment that the membrane
of the cell provides to the complex molecules inside, outside
of the cell in more complex life forms there are proteins that act
as molecular pumps, valves, regulators and gates that match
the complexity and functionality of equivalent machines made by
humans. They are equipped with the capability to distinguish among
the different molecules they pump and adjust the flow rates accordingly.
They thus determine the kind of molecules and how many enter and
exit the cell.(3)
If true, this is strong evidence that
complex life systems are not just the result of random
mutations but the result of purposeful design! ( also consider photosynthesis )
STABILITY: The most ancient fossils are of blue-green algae,
these being more than 3 billion years old. Thats right, the
same blue-green algae that exists today, in fact there is NO
evidence of change in blue-green algae over the billennia.(4)
How can anything be this stable? In the animal kingdom the shark also provides the same
evidence of excellence of design and
non-susceptibility to changes due to random mutation!
If true, this is strong evidence that
the life system was originated by an "Intelligent Designer"!

SUDDEN CHANGE: The fossil record indicates that for 3.3 billion
years of life history on Earth not much happened, there was only
single-celled creatures and long chains of similar single cells.
Then suddenly 543 million years ago species with specialized organs,
joints and appendages appeared. During this explosion of life, called the
"Cambrian explosion", more than 70 phyla appeared, all 30 phyla in existence today,
and including 40 or more now extinct. (5) All this probably occurring within
less than 3 million years (6) and in the following 540 million years no new phyla appeared.
Recent analysis of the marine invertebrate fossil record reveals that
biodiversity peaked soon after the onset of the Cambrian explosion and
has remained constant since then. Biodiversity
appears to have reached a ceiling level almost immediately after complex
animal life first appeared on the scene.(7)
If true, this rapid and varied change
in complexity of life forms defies a naturalistic explanation!

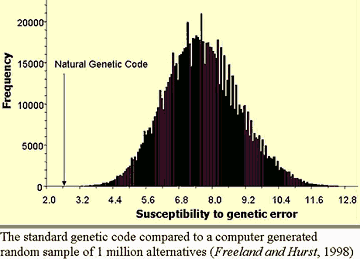
CODE OPTIMIZATION: The genetic code has a very unique error-minimization
capability. Researchers working with 1018
possible genetic codes (of the 1.4 X 1070
possible codes) with similar error-minimization
capabilities found that they mostly tended to group in
a normal distribution curve which they rated at code error
values of 3.2 to 12.4 . However, the universal genetic
code of our life system rated outside of this distribution
with a superior error code rating of 2.6 . (8)
"Only one in a million other possible codes is better". If a computer
was evaluating codes in order to select the best, to
come up with our universal genetic code it would have to
evaluate 1054 codes a second,(9) requiring a
very powerful super computer.
If true, this is strong evidence that
the code was selected by an "Intelligent Designer"!